- Determine the optimal timing of surgery following SARS-CoV-2 infection;
- Determine key global surgical indicators, such as postoperative mortality rates.
All hospitals are eligible to participate in this study, including hospitals that have not admitted SARS-CoV-2 infected patients.
Data collection will be on all surgical patients (children and adults) with and without SARS-CoV-2 infection. Hence, it is a unique opportunity to collect a snapshot of global surgical outcomes data for children in addition to assessing the impact of COVID-19. We encourage all to participate.
Sign-up from this week ready to start data collection for 1 week from the beginning of October.
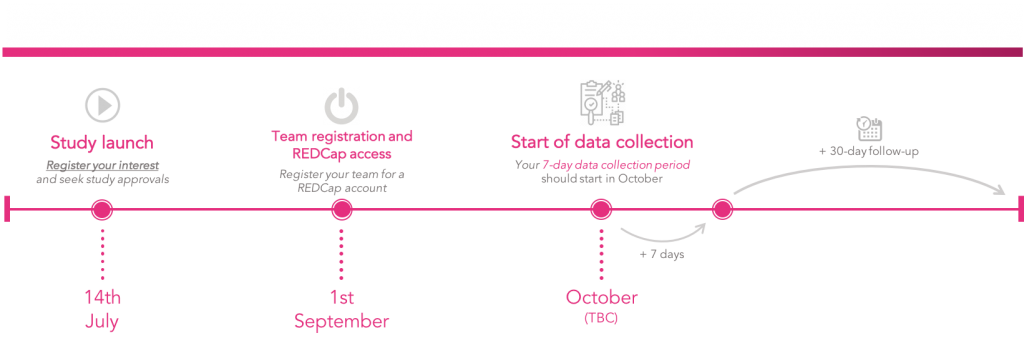
GlobalSurg | CovidSurg Timeline – accessed 16 July 2020, <https://globalsurg.org/surgweek>
For more information regarding registration or the protocol, kindly head to the CovidSurg website.




Leave A Comment